Material Transfer Agreement – Outgoing
MTAs describe the terms under which University researchers and outside researchers can share materials, typically for research or evaluation purposes. In cases when only human-derived data are being shared, a request for a Data Use Agreement (DUA) should be submitted. These agreements, along with equipment leases and loans, are handled by the Office of Research and Sponsored Projects.
Outgoing Material Transfer Agreements (MTA) govern the transfer of material from U-M to another organization.
If you are a U-M researcher interested in sending materials to an outside entity and you determine that you require a material transfer agreement (MTA) based on the criteria in the decision tree (see below), please complete an unfunded Agreement (UFA) request for an outgoing MTA in eResearch, under Proposal Management. For additional information on completing an UFA request, please refer to the UFA Quick Reference Card.
Most lab members can complete the online submission form (e.g., lab manager, research administrator, graduate student, post-doc, or Principal Investigator), but Principal Investigator (PI) information is required to submit the form and the PI will be notified of the MTA request.
If you are an outside entity interested in receiving material from a researcher at U-M, please contact U-M Innovation Partnerships at MichiganMTA@umich.edu and provide information about the materials and the recipient Principal Investigator.
If you are a U-M researcher interested in sending materials to an outside country, please contact MichiganMTA@umich.edu.
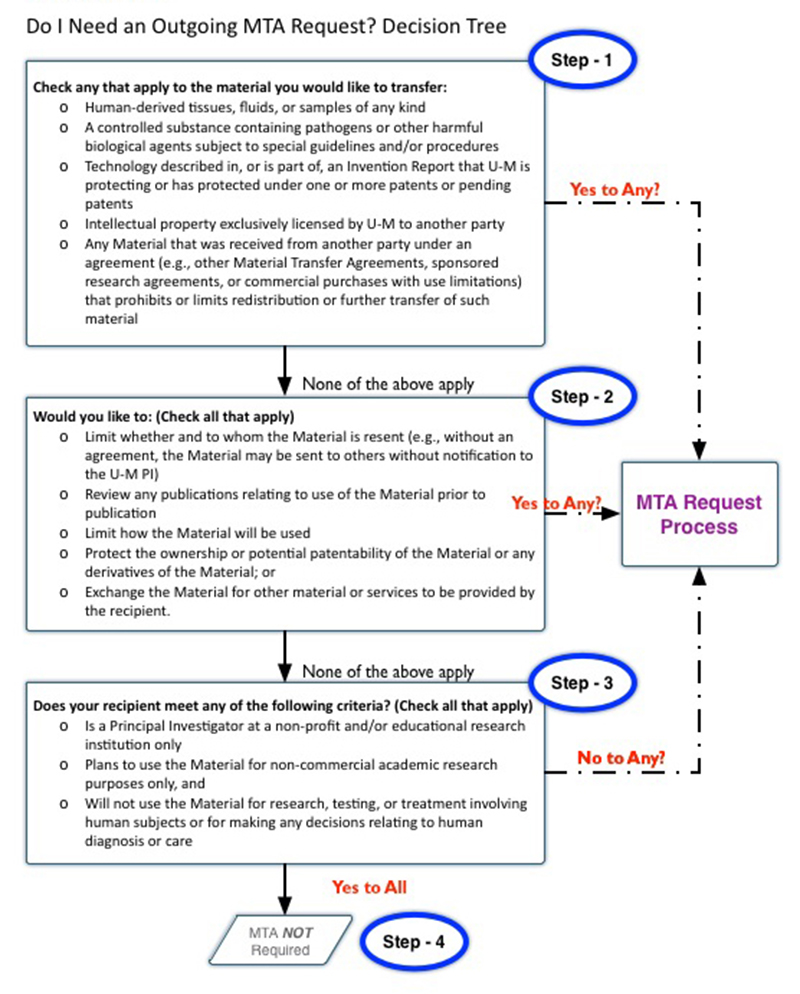
Decision Tree – Outgoing MTA
The following decision tree will help you to determine if an outgoing MTA request is needed.
For U-M Faculty
To process the MTA, U-M Innovation Partnerships will need the following information (and may request additional information, as necessary):
- Description and amount of material to be sent
- Name and contact information (including email) of Recipient
- Funding sources used to generate materials (e.g., PAF ID)
- IRB Informed Consent and Protocol sections that allow the transfer of human-derived materials
Sending Materials Outside of the United States
If you are shipping materials outside of the U.S., the materials may be subject to federal regulations, including export control laws, that define the conditions under which certain information, technologies, and commodities can be transmitted to individuals outside of the U.S. (including U.S. citizens that are outside of the U.S.) or, in the case of information, to certain foreign nationals within and outside of the U.S. Innovation Partnerships can review with you the conditions that may trigger review of the intended transfer under U.S. export control laws and regulations.
Processing an Outgoing MTA
- An eligible individual completes the Unfunded Agreement (UFA) request for the outgoing MTA in eResearch, under Proposal Management. An eligible PI signs off and in some cases, Unit approvers may sign off as well. For additional information on completing an UFA request, please refer to the UFA Quick Reference Card.
- U-M Innovation Partnerships reviews the MTA request and supporting documents, and then contacts the PI and/or contact person designated on the form if further information is necessary.
- U-M Innovation Partnerships drafts an appropriate outgoing MTA and sends it to the outside entity.
- If necessary, U-M Innovation Partnerships negotiates the MTA terms.
- Upon completion of the negotiation (if necessary), the outside entity signs the MTA and sends it to U-M Innovation Partnerships for final approval.
- U-M Innovation Partnerships notifies both parties that the materials can be transferred and sends a copy of the final agreement for their records.
Agreement Negotiations
Negotiation is often the most protracted part of the MTA process and the timeline of MTA processing depends on the extent of negotiation, the timing of the outside entity getting back to us, and the resolution of any compliance issues that need to be resolved prior to approval. You may check the status of your MTA using the Unfunded Agreement (UFA) section in the eResearch Portal.
FAQ
Should I submit an MTA request if I’m paying a third party to analyze some samples as a service and not for the third party’s research purposes?
No, these types of fee-for-service arrangements are handled by Procurement and you should contact them.
Should I submit an MTA request if I am only receiving or sending data without any physical materials?
No, in this case you will need to submit a request for a Data Use Agreement (DUA) through eResearch, which will be processed by ORSP.
Do I need to submit a request for an MTA if I am receiving an equipment loan?
No, you should contact ORSP with this request.
Can I charge a fee for providing samples to a non-profit entity?
As part of a non-profit institution supported by federal funding, U-M researchers may not charge for their materials to other non-profit institutions per NIH sharing policy. However, you can be reimbursed for all expenses directly incurred by the laboratory as a result of fulfilling the transfer. These include preparation of chemicals or reagents, portion of the cost of maintenance of an animal colony, technician time required to prepare materials and paperwork, along with all shipping costs associated with the transfer. If you have a question about whether an expense is reimbursable, please contact MichiganMTA@umich.edu.
Can I charge a fee for providing samples to a for-profit entity?
When transferring materials to a company, U-M Innovation Partnerships can negotiate a fee-MTA where recipient pays a one-time fee upon entering into an agreement. In addition, depending on the intended use of the materials by the company, U-M Innovation Partnerships can enter into a license agreement. To learn more about different options available to you, please contact a licensing specialist.
Are there any additional requirements for Howard Hughes Medical Institute (HHMI) Investigators?
Yes, in addition to U-M policies, investigators that are employed by the Howard Hughes Medical Institute are also subject to HHMI policies for sending materials that meet the above criteria. Please reference the following: HHMI Policies Regarding MTA.
Can I submit an outgoing MTA request for human-derived materials if the IRB study that covers them has a pending amendment that directly impacts their collection and/or transfer?
Yes, you can still submit an MTA request for transfer of these materials. However, the MTA will not be finalized until this amendment is approved by the IRB.
I would like to have an open-ended collaboration with a scientist at a non-profit entity where I will send materials now, and later her/she will send me results/data/materials that will be determined over the course of the research. Can I submit an MTA request for such an open-ended collaboration?
No, this is not generally done by U-M Innovation Partnerships. A two-way agreement can be formulated, but both parties have to be clear upfront on the materials to be transferred. U-M Innovation Partnerships does not discourage such open-ended collaborations, but in these cases it will be easy to submit a request for an incoming MTA when your collaborator is ready to send you materials.
For More Information
MTAs are administered by U-M Innovation Partnerships. Intellectual property rights can be endangered if materials are used without a proper MTA. For help with any MTA matter, contact John Corthell.

